|
Syrup From Different Cultivars Typically, the extension services recommend C.P. 36-111, C.P. 52-48, and C.P. 67-500 as cultivars for syrup production; the University of Florida also recommends Louisiana Ribbon, Louisiana Purple, and Louisiana Striped. I have observed that excellent syrup is also produced from C.P. 29-116 (Yellow Gal), from a large soft purple variety (perhaps the purple variety that Wiley (1902) described or (?) perhaps Louisiana Purple (said by Yoder (1925) to be the prevalent syrup cultivar in south Georgia and north Florida) or C.P. 31-511, a chewing variety) and from others. Ones selection of a cultivar is, in the end, a matter of personal preference, tradition, and adaptation to a particular region. Notwithstanding, it is instructive to read Broadhead and Zummo. They indicate that a suitable variety should produce a high-quality syrup with the following characteristics: light amber color; mild, sweet flavor typical of sugarcane syrup; no colloidal sedimentation, or sediment confined to a trace of small particles near the top of the container; no crystallization or, if present, limited to a few small crystals. In other places within the same bulletin, they discuss other desirable cultivar traits that primarily concern production (e.g., disease resistance, good stubbling, resistance to lodging, sufficiently early maturity, large-diameter soft stalks for efficient juice extraction, high brix, and high yield.). Undoubtedly, readers will take exception to some of the characteristics embraced by Broadhead and Zummo, as I do. I did not have the resources or desire to conduct a professional variety trial, but I was interested in making comparisons for my own purposes, implying that it should be grown on my own soil. In the end, I found, as expected, that the three cultivars all have merit. My final impressions were based on discussions with knowledgeable growers, samples of different syrups, and a cursory trial of my own for color and taste, which I will report here. The cultivars (C.P. 36-111, C.P. 52-48, and C.P. 67-500) were grown side-by-side; fertilization was primarily by a low-nitrogen formulation used for tobacco. Syrup from each cultivar was produced on the stovetop in a large stainless steel pot, a process that required about 2 hours per batch. Syrup was skimmed during processing and the final product was strained through four layers of cheesecloth as in some large-scale productions. As a reference, syrup (2002) made from a mixture of these three cultivars plus a purple variety was prepared in the traditional way in a cast-iron kettle. Color Without question, legions of sugar chemists throughout the world must know the precise molecular nature of the coloring matter in syrup because they must deal with the same compounds when they partition massecuite into crystals and molasses. Surprisingly, however, I could find very little when I searched exhaustively in the public domain (Agricola [the National Agriculture Librarys electronic data base of citations] and ISIs Web of Knowledge [available only by subscription]). The questions are two-fold, providing synergistic answers. What is the material, and how does one enhance it or diminish it? As it turns out, the coloring matter is not one single compound or simple. Tu and Kondo (J. Chromotography 81: 187-189) indicate two heterogenous mixtures. One is a brown mixture with a Mr ranging above 5000; the other is a brown to yellow mixture ranging between 150-350. These authors cite their previous work indicating that leaves and the upper portion of the cane is the major source of the high Mr mixtures. Corroboratively, Yoder (USDA Bulletin 1034, 1925) indicates that immature cane cannot produce clear and light-colored syrup. I speculate that this higher molecular weight mixture consists of higher order polyphenols and early chemists (Schneller, Louisiana Bulletin 157, 1916) showed that immature portions of cane contain the bulk of polyphenolics. Polyphenolics can impart color on their own (as in browning of a cut apple), but they can also react with other substances, notably iron, to impart color. Different cultivars vary in iron content (Fort and McKaig, USDA Technical Bulletin 688, 1939) and intense milling increases the iron content of the juice (Fort and McKaig op. cit.). The short story seems to be that immature cane and high extraction rates, as mentioned by Paine and Walton (USDA Bulletin 1370, 1925), contribute to color. Paine and Walton (op. cit.) as well as Dale and Hudson (USDA Bulletin 921, 1920) warn against particles of bagasse in the juice, claiming that it might scorch during syrup production and impart color. Spencer (Florida Ag Bulletin 118, 1913) particularly stresses removing particles from the juice (wire screen, cloth strainer, muslin, then woolen blanket). Arkansas Bulletin No. 22 (1892) even goes a step further and suggests filtration of the juice through a 5-foot box of straw in order to produce a light-colored syrup. Indeed, Stockbridge (Florida Ag Bulletin No. 44, 1898)) evaluated a range of filtering substances (cotton-lint, shaving, . . .) and recommended Spanish moss as the best material to remove coloring matter from the juice. Texas Ag Bulletin 68 (1903) also used Spanish moss (a half barrel, washed and scalded each day) to remove color from juice. With all this emphasis on cleaning the juice to produce light-colored syrup during the heyday of syrup making, it is a bit odd that most present-day syrup makers pay little attention to this step. Absolute and strict removal of all cell-wall debris will also prevent formation of some colloids in the syrup (mainly from pectin and pentosans (hemicelluloses, see Sugar Technology Series No. 1, Imperial College of Tropical Agriculture, Trinidad, 1929). Caramelization (burning sugar) is another source of color (Dale and Hudson, op. cit.). Caramelization can be diminished by avoiding hot surfaces with dried juice or semi-syrup.
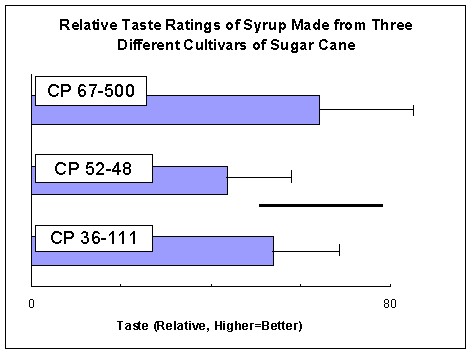
Taste Six subjects, representing both genders and two ethnic groups, evaluated the taste of the syrup samples in a blind test conducted in isolation. All subjects were consumers of sugar-cane syrup and professed a fondness for it. Each subject rated five unidentified syrup samples (one each of the four syrups plus a duplicate of one of the samples) on a 100-point scale, with 100 points being the best. Scores for each subject were normalized to 50, and then the normalized scores for each syrup were evaluated (Figure 2). The dark bar in Figure 2 is the standard deviation of the differences in scores given to the same syrup by the same subject; interestingly, one subject rated the very same syrup as the best and the worst of the lot and the difference between those scores was a whopping 53 points. In broad terms, this means that samples that did not differ in score by more than 18 points were probably indistinguishable to the subjects. Syrup made from C.P. 67-500 had the highest average score, 64, and was better (p~0.05, t-test, 1-tailed, equal variance) than that made from C.P. 52-48, which averaged 44 points. The data suggested that C.P. 36-111 syrup is also better than CP 52-48 syrup (p~0.12). Syrups of C.P. 36-111 and C.P. 67-500 were indistinguishable statistically. In conclusion, C.P. 36-111 produced syrup slightly lighter than C.P. 67-500 and these two cultivars were favored in a small tasting by sugar-cane syrup aficionados. |






 Slide 1
Slide 1