|
As succinctly put by Bhandari & Hartel (Journal of
Food Science, 67: 1797-1802), the control of crystallization of sugars is a
major concern, being important to the texture (e.g., of some candies) and
appearance (e.g., icing) of foods. In some foods, such as honey, crystals are
perceived as a natural and favorable attribute in some cultures, such as
Brazil's, and as a defect in other cultures, such as ours. Yet, in other
cultures, such as parts of Europe, deliberate and controlled crystal formation
("creamed honey") is widely practiced. Generally, however, crystals in syrups
are thought undesirable, and indeed, may negatively
affect storage because the remaining liquid is less concentrated. Still, in
my opinion, it is important to note, as seen by the example of honey, that
markets might respond to a non-liquid spread with the taste of sugar-cane syrup
or of sorghum syrup.
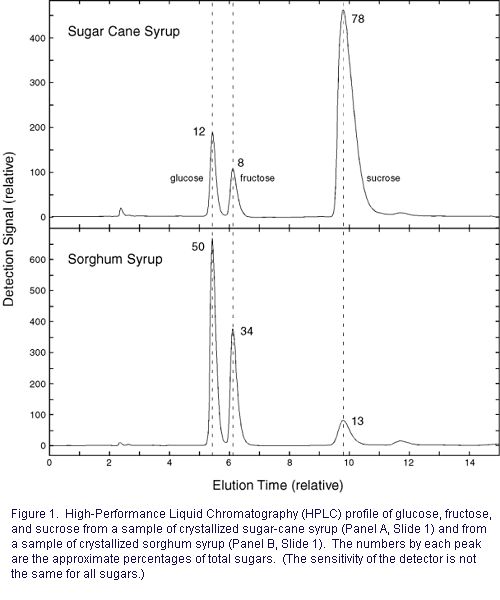
Crystal formation in sugar cane syrup is common and crystal formation occurs some in sorghum syrup as well (Panel A and B, respectively, Slide 1). Prevention of crystal formation in sugar-cane syrup is theoretically straightforward if in practice lacking details: conversion of sucrose to its constituent monosaccharides, glucose and fructose, will eliminate crystallization. This conversion may be effected by use of invertase or by acid + heat treatment. (The efficacy of invertase is discussed elsewhere; use of lemon juice during processing by one Mississippi sugar-cane syrup producer is known.) Consistently, the crystallized sugar-cane syrup sample (Panel A, Slide 1) had a high sucrose content (Fig. 1). (Sucrose is the least soluble of the three major sugars.) As indicated above, in all probability, this syrup would not have crystallized had the sucrose content been reduced. In some cases, reduction of the sucrose content by use of invertase has been advocated by extension personnel as a means to prevent sugaring in sorghum syrup. These sources recommend using half as much invertase as necessary for sugar-cane syrup. Crystallization can be a complex process, and crystals form in sorghum syrup that has low sucrose content, too (Fig. 1). In such a sample, invertase would not be effective, of course. Glucose is the most abundant sugar in this sample, and glucose crystallization is the main foundation for formation of creamed honey. (Essentially, the only sugars in honey are glucose and fructose. Honey high in glucose, such as that produced from certain floral sources like mustard, are the best for making creamed honey, and honey high in fructose, such as tupelo, could probably not be crystallized under the same conditions.) Unfortunately, there are no turnkey methods for conversion of glucose to fructose in syrup (although the concepts are in place to convert part of glucose to fructose, depending on the starting ratio of the two sugars.) The reader should note that I do not have any direct experience in producing sorghum syrup before putting a value on my thoughts for preventing crystallization if it is a problem: (a) determine whether sucrose is a cause of crystallization, or simply use invertase as a prophylactic. (b) avoid seeding. Crystals grow on "seeds," small surfaces, such as tiny crystals themselves. It is common practice to open liquid syrup and note that it crystallizes in days because seed crystals form as syrup concentrates on the container, especially near the spout, and forms seeds. When these seeds mix with the syrup, there is a cascading effect. (c) avoid storage under conditions that promote crystallization. That is, avoid time-proven empirical methods for making creamed honey. (d) reduce the brix, if feasible. I mention again in closing that I do not have specific expertise in the prevention of crystallization in sorghum syrup, and that these suggestions simply stem from my general knowledge of chemistry. I would be delighted to incorporate other thoughts into this essay, and am particularly seeking solid information that I could use to update this essay. |